

Here's a unique bioresearch application for Vydac's 302IC4.6 column, originally designed for separating inorganic ions in environmental analysis. The packing, a low-capacity quaternary amine anion exchanger based on high-purity 10µm large-pore silica, separates organic ions by a mixture of ionic and reversed-phase (hydrophobic) interactions.
The nucleotide separation shown in Figure 1 was performed with a mobile phase that is atypical by reversed-phase standards because it contains no organic solvent, thus simplifying disposal. Instead, the acetic acid used for pH adjustment takes the place of the organic solvent in facilitating the reversed-phase portion of the mixed-mode separation.
As indicated by the elution order, ionic interactions due to the negative
charges of the phosphates appear to play a major role, separating mono-,
di-, and tri-phosphate nucleotides as groups. Within each group, differing
properties of the bases result in separation of individual nucleotides. Hydrophobic
interactions vary with size of the heterocyclic ring structure and tend to
cause purine nucleotides to be retained longer than pyrimidine nucleotides.
However, the bases also carry positive charges at the mobile-phase pH, thereby moderating ionic retention. Uracil is the least basic, reflecting its lack of an amino substituent on the heterocyclic ring. This accounts for longer retention of uridine nucleotides and their apparent anomalous elution between the corresponding purine nucleotides.
The 1:1 proportion of dibasic and monobasic phosphate salts in the mobile
phase was important in achieving conditions for this separation. Using only
dibasic phosphate eluted all peaks too early, probably because more acetic
acid was needed to adjust the pH. Using exclusively monobasic phosphate
eluted all peaks much later.
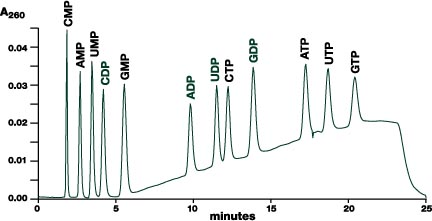
Figure 1. Separation of twelve common nucleotides on Vydac ion chromatography
column.
Column: Vydac 302IC4.6, 4.6mmID x 250mmL. Flow rate: 2.0 mL/min.
Detection: 260nm. Mobile phase: A = NaH2PO4/Na2HPO4
(1:1 molar ratio) at 25mM total concentration in water, adjusted to pH 2.8
with acetic acid. B = NaH2PO4/Na2HPO4
(1:1 molar ratio) at 125mM total concentration in water, adjusted to pH
2.9 with acetic acid. Gradient: 0% B for 2 minutes, then linear from
0% to 100% B in 17 minutes, hold 100% for 2 minutes, return to 0% B in 0.1
minute. Sample: 3µL containing 0.25µg of each nucleotide.
| Vydac Cat. # | Description |
|---|---|
| 302IC4.6 | Column, Ion Chromatography, Quarternary Amine, 10µm, 4.6mm ID x 250mm L |
| 302IC4.610 | Column, Ion Chromatography, Quarternary Amine, 10µm, 4.6mm ID x 100mm L |
Other analytical and preparative column dimensions available upon request.
Last Updated: 08/20/21
"Vydac" is a trademark of The Separations Group.